Draft with context
Generate clinical content from governed study records, evidence, rationale, and approved design decisions.
Solutions Medical Writing
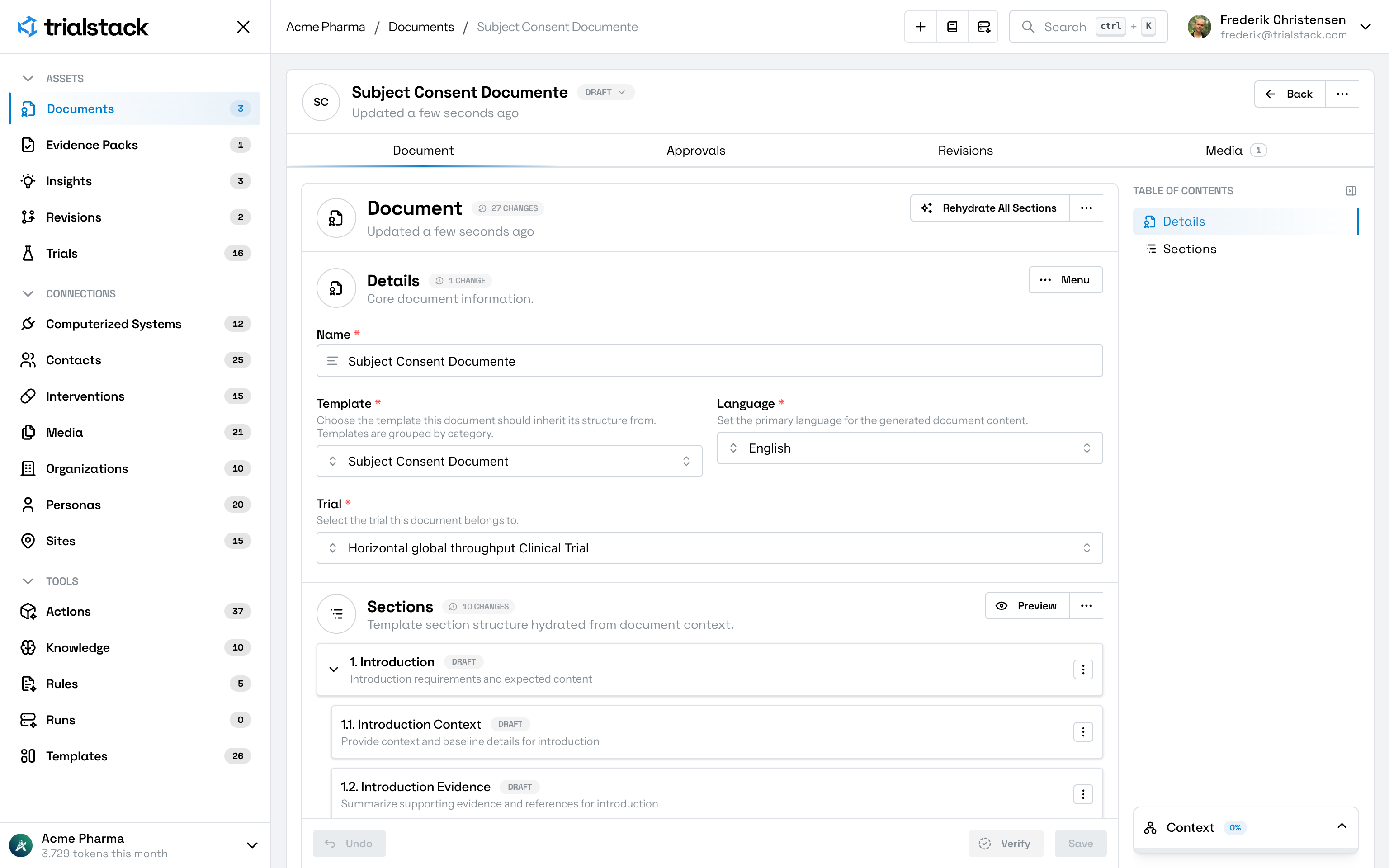
Draft, revise, and review clinical documents from structured study decisions, source evidence, and approval history instead of rebuilding context across files, comments, and disconnected authoring tools.
WRITE AGAINST THE RECORD
Keep protocols, synopses, ICFs, SAP inputs, and submission materials connected to the evidence, rationale, comments, and approvals behind them.
Generate clinical content from governed study records, evidence, rationale, and approved design decisions.
Move comments, revisions, approvals, and handoffs through visible paths built for regulated writing work.
Update documents while keeping changes connected to the decisions, source material, and rationale that shaped them.
Maintain a traceable record of versions, changes, review activity, and approval decisions across the content lifecycle.
Built on trusted standards
Structured around the standards that guide modern clinical development.
Pricing
Start with one active trial, then scale into multiple studies, larger teams, higher AI usage, stronger governance controls, and priority support as your study-startup work grows.
Ignite
For small teams launching a first trial who need core governance, evidence, and document controls.
Accelerate
For teams managing multiple active studies that need more capacity, stronger governance, and faster support.
Scale
For enterprise teams running larger study portfolios with custom limits, identity controls, and dedicated support.
Get started
Talk through your trial design, evidence review, document generation, and start-up workflows, then choose the Studio, Loop, or Catalyst path that fits your team.