Centralize source material
Keep literature, guidelines, prior submissions, supporting documents, and study artifacts connected inside the same governed workspace.
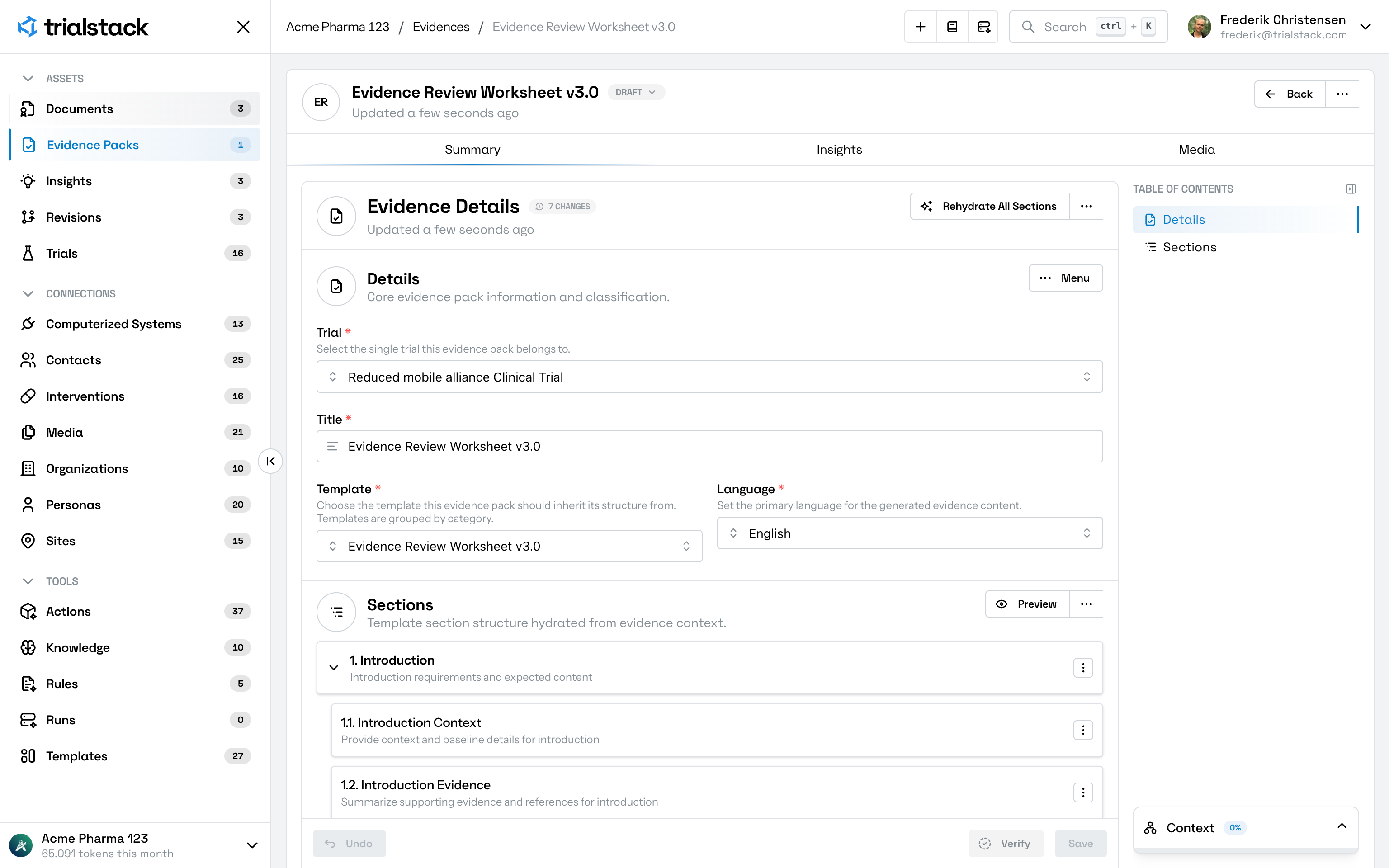
Solutions Evidence Synthesis
Turn literature, prior documents, reviews, and study artifacts into linked evidence packs and structured summaries without losing source context, review history, or downstream traceability.
SYNTHESIZE WITH TRACEABILITY
Bring source review, evidence packaging, structured summaries, and reusable rationale into one connected workflow instead of stitching evidence together by hand.
Keep literature, guidelines, prior submissions, supporting documents, and study artifacts connected inside the same governed workspace.
Produce reusable evidence outputs in formats teams can review, compare, and carry into protocol design, documents, and downstream decisions.
Put expert validation into the synthesis path so downstream teams inherit reviewed evidence, not rough notes or unsupported summaries.
Keep every evidence output connected to the source material, rationale, review activity, and decisions it supports.
Built on trusted standards
Structured around the standards that guide modern clinical development.
Pricing
Start with one active trial, then scale into multiple studies, larger teams, higher AI usage, stronger governance controls, and priority support as your study-startup work grows.
Ignite
For small teams launching a first trial who need core governance, evidence, and document controls.
Accelerate
For teams managing multiple active studies that need more capacity, stronger governance, and faster support.
Scale
For enterprise teams running larger study portfolios with custom limits, identity controls, and dedicated support.
Get started
Talk through your trial design, evidence review, document generation, and start-up workflows, then choose the Studio, Loop, or Catalyst path that fits your team.